All About TORCH Infections
 The ToRCHeS are a group of infections that can be passed from the pregnant mother to her fetus through the placenta. They can infect anytime during gestation, and sometimes during the time of delivery as well. Since they can cause mental retardation, morbidity, and even death, they are important to recognize and prevent. So far in my pediatric rotation, I have seen a case of it, and so it is something we will see more as doctors.
The ToRCHeS are a group of infections that can be passed from the pregnant mother to her fetus through the placenta. They can infect anytime during gestation, and sometimes during the time of delivery as well. Since they can cause mental retardation, morbidity, and even death, they are important to recognize and prevent. So far in my pediatric rotation, I have seen a case of it, and so it is something we will see more as doctors.
First off, ToRCHeS stands for:
- To = Toxoplasma
- R = Rubella
- C = Cytomegalovirus
- He = Herpes Simplex Virus
- S = Syphilis
Althernatively, it could also be spelled as TORCH, in which the O stands for “other.” The “other” organisms that cause perinatal infections are Parvovirus B19, Varicella Zoster Virus, Syphilis, and HIV. However, here, I’ll be talking about ToRCHeS.
Toxoplasma
Caused by the protozoan called toxoplasma gondii, Toxoplasma is very common parasite found in humans. In the US, up to 15% of people have tested seropostive for it. In other countries like in France, it is 90% and in Germany it is 50%. It may be very common, but most people who are immunocompetent are asymptomatic. You usually only find symptoms of the infection when people become immunocompromised, like in HIV patients.
TRANSMISSION
Toxoplasma have their sexual cycles in cats and asexual cycles in other mammals and birds. Cats often get them by eating rodents, birds, and other small animals outside. Toxoplasma then reproduces in the cat’s GI tract, producing lots of oocytes, which then gets released in the cat’s feces. When other animals outside get contaminated with these feces, then they too catch the infection. Toxoplasma spreads through the blood and eventually encysts in tissue cells permanently (but contained by our immune systems). While humans can get it through handling cat litter, the major cause of transmission in the US and Europe is actually through eating meat containing the cysts, particularly pork, venison, and lamb.
CONGENITAL PRESENTATION
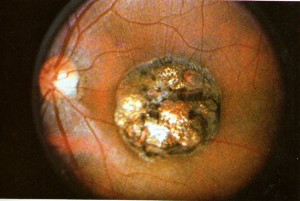
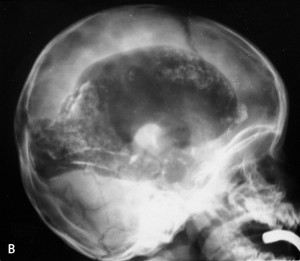
When a pregnant woman gets infected with toxoplasma, there is a chance that the infection can be passed to the fetus via the chorionic villi in the placenta. When this happens, you may see the classic congenital toxoplasma triad of…
- Chorioretinitis
- Hydrocephalus
- Intracranial calcifications
Even with the infection, a good majority (67% of infected fetuses) are born with no symptoms. 15% are born with retinochoroiditis and 10% have intracranial calcifications. The symptoms become more severe when the infection happens early in pregnancy.
DIAGNOSIS
So how do you screen for congenital toxoplasma? You do antibody titers, looking for T. gondii-specific IgM in the pregnant mother, then do an amniotic fluid DNA PCR to detect the infection in the fetus.
TREATMENT
If the mother is confirmed to be infected with toxoplasma, then treat the mom with spiramycin. If both the baby and the mom are infected, then treat the mother with pyrimethamine and sulfonamide, which will cross the placenta and treat the fetus as well. But because pyrimethamine and sulfonamide have the adverse effect of bone marrow suppression, you would also want to treat with leukovorin. Also because there is risk for retinitis, you’d also want to give to refer the baby for an ophthalmic examination for risk for retinitis.
PREVENTION
To prevent congenital toxoplasma in the fetus, here are some tips for pregnant women:
- Avoid handling cat litter, particularly cats who have access to the outdoors where they may eat infected rodents and small birds.
- Cook meats very well. Remember this is the most common cause of transmission in the US.
- Wear gloves if you are going to be working in the garden or will be handling soil which may potentially have toxoplasma.
- Wash garden vegetables very thoroughly, to prevent any contamination.
Rubella
Rubella is also known as “German Measles.” Today, it is rare in the US because of the widespread administration of MMR vaccine. Rubella cases in the US mainly happen in mothers from countries who did not have an MMR vaccination program.
TRANSMISSION
Rubella is spread through respiratory droplets, and the mother who gets it will show upper respiratory tract symptoms, rash that starts on the trunk, lymphadenopathy, and arthritis. Like all other TORCH infections, the mother may also be asymptomatic yet still have the risk of transmitting the infection to fetuses.
CONGENITAL PRESENTATION
In congenital rubella, you see a triad of:
- Patent ductus arteriosus
- Cataracts
- Deafness
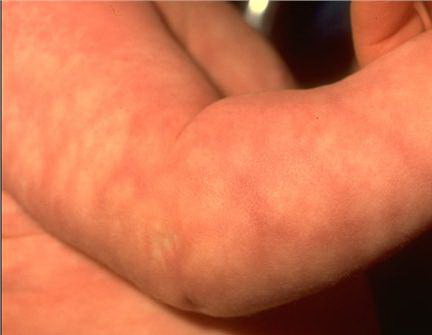
The baby is often born with intrauterine growth restriction (IUGR), meaning they are restricted from growth in the placenta. It is often symmetric, meaning both the head and the body are restricted from growing, and this causes the baby to be born with a small head (microcephaly), and therefore mental retardation. The baby may also have what is called a “blueberry muffin rash.”
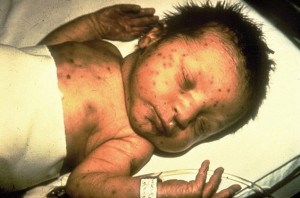
DIAGNOSIS
To diagnose, you test the maternal and fetal serum for antibodies against rubella. You can also detect the virus in the amniotic fluid as a way for diagnosis.
TREATMENT
Unfortunately there is currently no antiviral treatment available for congenital rubella, and the only thing you can do is give the baby supportive therapy.
PREVENTION
Rubella, however, is an easy disease to prevent, and anyone who has not been vaccinated with MMR should consider it before getting pregnant. For those women who do get the vaccine, they should wait at least 28 days before attempting to get pregnant, since MMR is a live vaccine and your body needs time to build up those antibodies against it.
Cytomegalovirus
Cytomegalovirus (CMV) is another very common infection. In fact, up to 60% of adults in the US have tested seropositive for it, and like toxoplasma, most people’s immune systems are strong enough to make them asymptomatic.
TRANSMISSION
CMV is transmitted via bodily fluids, which includes sexual contact, organ transplants, blood transfusions, breast milk, saliva, urine, and transplacentally from mother to fetus. Usually it can transmit from the mother to fetus when the mother has either a primary infection or reactivation of the CMV during pregnancy. The mother may have heterophile negative mononucleosis, presenting with fever, sore throat, and fatigue. Like other TORCH infections, mothers who do not show signs of the infection can still be at risk of passing the infection to their fetuses.
CONGENITAL PRESENTATION
Congenital CMV infects up to 40,000 newborns annually in the US but the majority are asymptomatic. Only 5-20% of those infected show any signs or symptoms.
For those who are born with symptoms, you may see intrauterine growth restriction (leading to mental retardation and microcephaly), jaundice, seizures, periventricular calcifications (in brain on CT), hearing loss, petechial rash, pneummonia, hepatosplenomegaly, or psychomotor retardation.
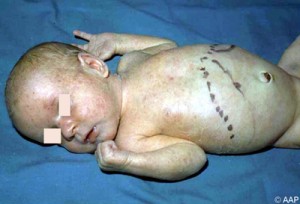
DIAGNOSIS
You can diagnose the doing CMV titers (part of TORCH screen) on the newborn baby and confirm with viral cultures from urine or saliva samble from the baby.
TREATMENT
Unfortunately, like Rubella, there is currently no specific treatment for congenital CMV. Physical and supportive therapy is the only thing you can do at this point, like giving hearing aids, special education, etc.
PREVENTION
To prevent congenital CMV, the pregnant mother should wash hands thoroughly after touching diapers or saliva, and avoid any contact with bodily fluids, particularly from young children who are at more risk of getting CMV. Women who are confirmed to be infected should get ganciclovir treatment to decrease the risk of spread to the fetus.
HERPES SIMPLEX VIRUS
Herpes simplex virus (HSV) comes in two strains, HSV 1 (oral herpes), and HSV 2 (genital herpes). HSV 1 can also be found in genital herpes and vice versa, but in terms of congenital HSV, HSV 2 is the more common one you’ll see.
TRANSMISSION
HSV is transmitted via skin or mucous membrane contacts, such as during birth through an infected genital tract.
Mothers that have an HSV outbreak often have vesicular lesions on their genitals. Like many TORCH infections, mother who have genital herpes often may not show signs of an outbreak, but still have the risk of transmitting it to the fetus during vaginal birth.
NEONATAL PRESENTATION
Neonatal HSV can present in three ways:
- Locally on skin, eye, and mouth, known as SEM. Neonates with this presentation have the best survival and usually have no mental retardation or developmental delays.
- Central nervous system infection. You see meningoencephalitis, seizures, bulging anterior fontanel, poor feeding, and irritability.
- Disseminated. This is the worst presentation and you see sepsis, shock, and respiratory failure. You also often see the CNS and local infections as well.
DIAGNOSIS
To diagnose on a neonate, you can take a swab sample from the conjunctivae or oropharynx of the neonate and do a viral culture to detect the virus. You could also detect viral DNA via PCR.
TREATMENT
Neonates who have a confirmed diagnosis of HSV should be treated with a full course of intravenous acyclovir. Babies with SEM must be treated for at least 14 days. For those with CNS or disseminated infections, at least 21 days of treatment is needed.
PREVENTION
If you suspect HSV in the mother, or if the mother has a history of HSV, then you should recommend doing a c-section to prevent the fetus from passing through a potentially infected vaginal canal.
Syphilis
Syphilis is caused by the spirochete bacterium called Treponema pallidum.
TRANSMISSION
In adults, it is transmitted via sexual contact, and people usually go through three phases of the disease:
- Primary syphilis — painless chancres
- Secondary syphilis — disseminated rash, condylomata lata
- Tertiary syphilis — neurosyphilis, gummas, tabes dorsalis.
CONGENITAL PRESENTATION
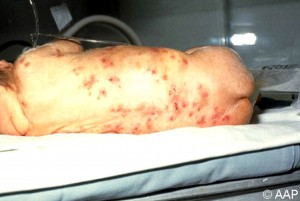
Unlike other TORCH infections where the infected patient is often asymptomatic, in congenital syphilis, 40% of fetuses get hydrops fetalis and do not make it past birth. For those babies that do get born, most of them get symptoms.
- Early symptoms (before 2 yrs): Early on, you will find failure to thrive, anemia, maculopapular rash, hepatosplenomegaly, periostitis, and rhinitis.
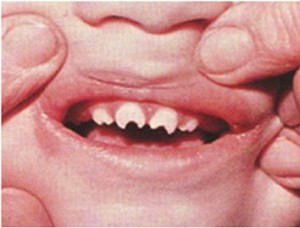
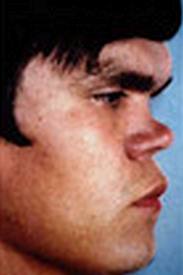
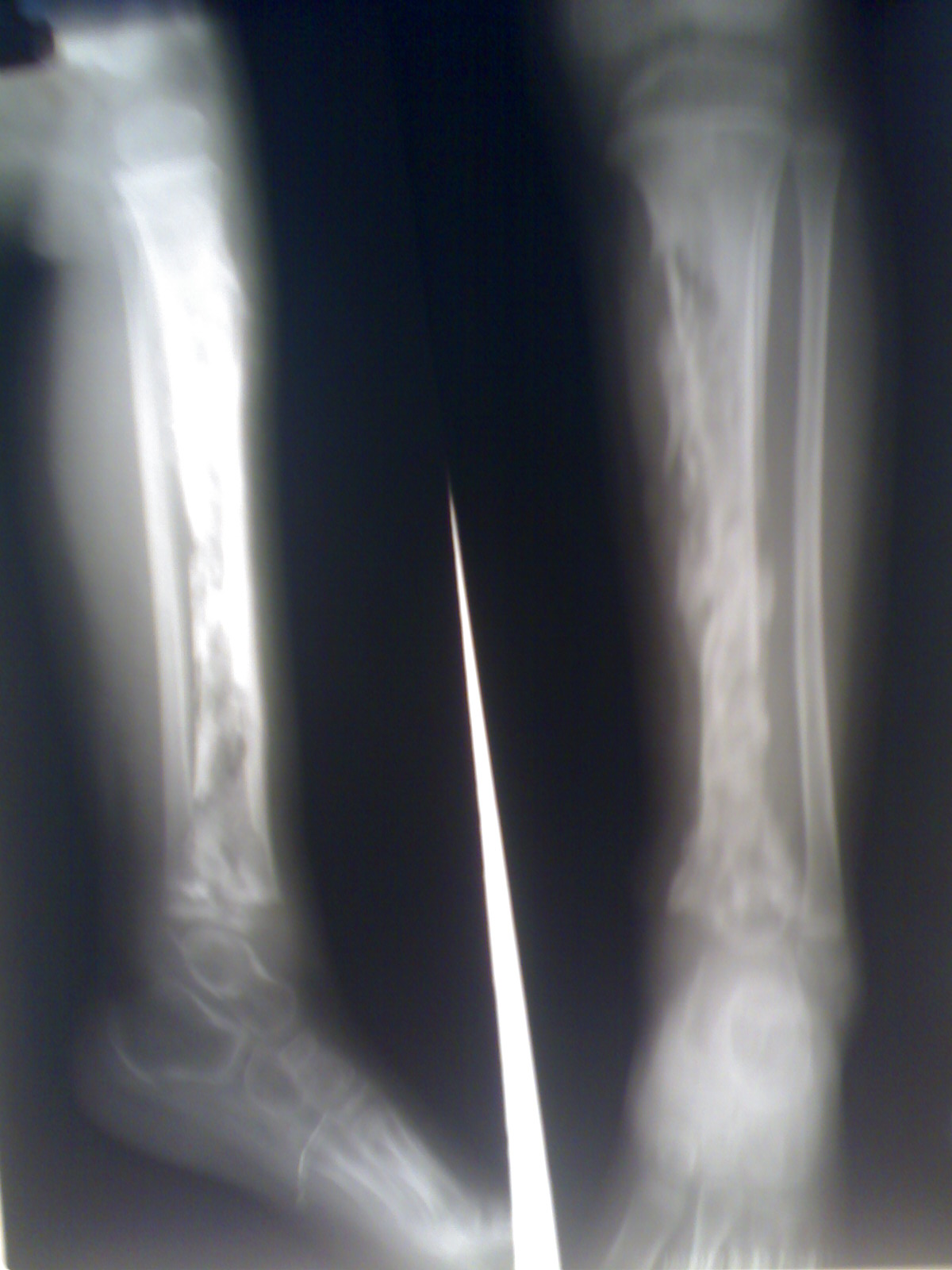
- Late symptoms (after 2 yrs): Later on, the patients may develop Hutchinson teeth (notched teeth), saddle nose (from bridge of nose breaking), saber shin (anteriorly-curved tibia), and short maxilla.
DIAGNOSIS
You can diagnose with an RPR or VDRL test, followed by FTA-ABS to confirm. You could also do dark field microscopy to detect the bacterium, which is a more accurate test, especially in primary syphilis.
TREATMENT
If the neonate tested positive for syphilis, treat with parenteral penicillin G for 10 days. If the neonate tested negative but the mother is postive for untreated syphilis, then treat the baby with a dose of IM benzathine penicillin.
PREVENTION
To prevent congenital syphilis, screen the expectant mother for syphilis and treat her with penicillin G to minimize transmission to the fetus.
Resources: Medscape, UpToDate, First Aid for USMLE Step 1, Pub Med Health












This was incredibly detailed and helpful, thank you!
thank you so much for this helpful info. excellent work!!